I had a brain-blast last night. I haven't *fully* confirmed anything yet, but I've got a theory.
I had issues with ammoniacal emulsions fogging a lot, and I always used erythrosine for them too. Regular ammonia-bromide emulsions seemed fine, at the time. But later on, I noticed that when testing gold/sulfur sensitization, I would get some pretty bad fog that would absolutely nuke the emulsion as the sensitization carried out.
I started to suspect my "safelights" might be leaking a little bit of green. I just use cheapy wifi RGB bulbs set to red. They never seemed to give me any problems in the past, but it's been a few years since I worked with paper or anything "normal". I started keeping them at 1% while taking samples, and turning them off entirely in-between, and the mystery fog disappeared.
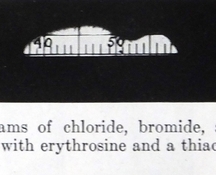
Last night I was curious, and held up a $10 spectroscope to each bulb, and sure enough there was a good amount of yellow light in all of them, and a couple even had some green light visible.
None of this was scientific enough to really say the bulbs are the 100% culprit here, as I changed a bunch of other small stuff with my workflows at the same time. But it would make a lot of sense if this was the case.
I ordered some "deep red" LED strips off of eBay last night, and I'll update when I can test those out.